The growing interstitial lung disease market is supported by the widespread use of foundational and supportive treatments such as corticosteroids, immunosuppressants, biologics, and adjunct therapies like cough suppressants. At the same time, the introduction of advanced pipeline candidates, including seralutinib (Gossamer Bio/Chiesi Farmaceutici), L606 (Liquidia Corporation), N-115 (Emphycorp), efzofitimod (aTyr Pharma), and others, is expanding the treatment landscape by addressing multiple disease pathways, thereby driving sustained market growth and ongoing therapeutic innovation.
New York, USA, April 08, 2026 (GLOBE NEWSWIRE) — Interstitial Lung Disease Market to Witness Accelerated Growth at a CAGR of 8.7% During the Forecast Period (2026–2036) Due to the Launch of Emerging Therapies | DelveInsight
The growing interstitial lung disease market is supported by the widespread use of foundational and supportive treatments such as corticosteroids, immunosuppressants, biologics, and adjunct therapies like cough suppressants. At the same time, the introduction of advanced pipeline candidates, including seralutinib (Gossamer Bio/Chiesi Farmaceutici), L606 (Liquidia Corporation), N-115 (Emphycorp), efzofitimod (aTyr Pharma), and others, is expanding the treatment landscape by addressing multiple disease pathways, thereby driving sustained market growth and ongoing therapeutic innovation.
Recently published Interstitial Lung Disease Market Insights report includes a comprehensive understanding of current treatment practices, interstitial lung disease emerging drugs, market share of individual therapies, and current and forecasted market size from 2022 to 2036, segmented into leading markets [the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan].
Interstitial Lung Disease Market Summary
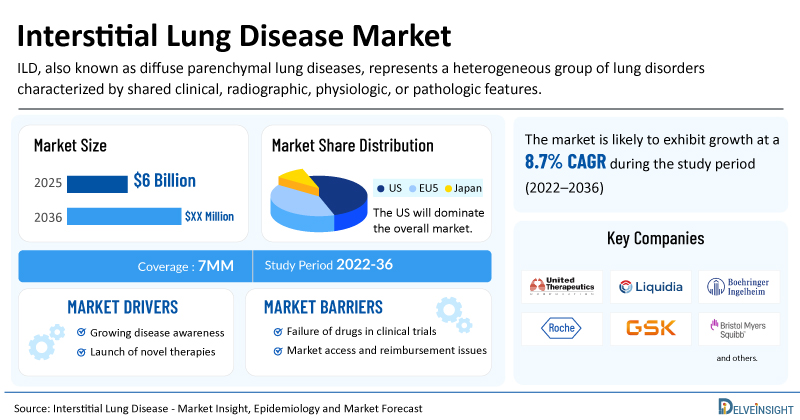
- The market size for interstitial lung disease was found to be USD 6 billion in the 7MM in 2025.
- The United States accounted for the largest interstitial lung disease treatment market size in 2025, i.e 83%, compared to other major markets, including the EU4 countries (Germany, France, Italy, and Spain), the United Kingdom, and Japan.
- The total number of diagnosed prevalent cases of ILD in the 7MM is ~1.3 million in 2025.
- Key interstitial lung disease companies, including GSK, Insmed, Gossamer Bio, Chiesi Farmaceutici, Liquidia Corporation, Bristol-Myers Squibb, Emphycorp, aTyr Pharma, PureTech Health, Endeavor Biomedicines, Avalyn Pharma, and others, are actively working on innovative interstitial lung disease drugs.
- Some of the key interstitial lung disease therapies in clinical trials include Belimumab (BENLYSTA), Treprostinil Palmitil Inhalation Powder (TPIP), Seralutinib, L606 (Treprostinil Liposome Inhalation Suspension), Admilparant (BMS-986278), N115 (Nonsteroidal Nasal Spray), Efzofitimod, Deupirfenidone (LYT-100), Taladegib (ENV-101), AP01 (Inhaled Pirfenidone), and others. These novel interstitial lung disease therapies are anticipated to enter the interstitial lung disease market in the forecast period and are expected to change the market.
- In 2036, among all the therapies for ILD, the highest revenue is estimated to be generated by JASCAYD (nerandomilast tablets) in the 7MM.
Discover which segment dominates ILD market @ https://www.delveinsight.com/sample-request/interstitial-lung-disease-market
Key Factors Driving the Growth of the Interstitial Lung Disease Market
- Rising Prevalence of ILDs: Increasing incidence of conditions such as idiopathic pulmonary fibrosis (IPF), connective tissue disease-associated ILD, and occupational lung diseases is expanding the patient pool globally.
- Advancements in Diagnostic Technologies: Improved imaging techniques (e.g., high-resolution CT scans), pulmonary function testing, and biomarker research are enabling earlier and more accurate diagnosis.
- Increasing Awareness and Screening: Enhanced physician awareness and patient education are leading to earlier detection and higher diagnosis rates.
- Launch of Emerging ILD Drugs: The dynamics of the ILD market are expected to chnage in the coming years due to the launch of emerging therapies such as Belimumab (BENLYSTA) (GSK), Treprostinil Palmitil Inhalation Powder (TPIP) (Insmed), Seralutinib (Gossamer Bio/Chiesi Farmaceutici), L606 (Treprostinil Liposome Inhalation Suspension) (Liquidia Corporation), Admilparant (BMS-986278) (Bristol-Myers Squibb), N115 (Nonsteroidal Nasal Spray) (Emphycorp), Efzofitimod (aTyr Pharma), Deupirfenidone (LYT-100) (PureTech Health), Taladegib (ENV-101) (Endeavor Biomedicines), AP01 (Inhaled Pirfenidone) (Avalyn Pharma), and others.
According to Ramandeep Singh, Senior Consultant of Forecasting at DelveInsight, the ILD therapeutic pipeline is undergoing a decisive shift from conventional anti-fibrotic and broad immunosuppressive strategies toward targeted biologics, novel small molecules, and advanced inhaled delivery platforms. Several late- and mid-stage programs now aim to address immune dysregulation, fibrotic signaling, and pulmonary vascular remodeling in a more mechanistically precise manner. BENLYSTA, developed by GSK, represents a shift toward B-cell-targeted therapy in autoimmune-driven interstitial lung disease, particularly systemic sclerosis–associated and connective tissue disease–associated ILD.
Interstitial Lung Disease Market Analysis
- Approved antifibrotic therapies are frequently linked to gastrointestinal, hepatic, and tolerability-related side effects, often necessitating dose adjustments or treatment discontinuation.
- Long-term daily oral administration further exacerbates adherence issues, especially among elderly patients or those with underlying autoimmune conditions.
- Additionally, inhaled prostacyclin therapies for ILD-associated pulmonary hypertension, such as TYVASO, introduce complexities related to device-based delivery.
- Revenue growth in the ILD market is increasingly driven by strong adoption of high-value, differentiated therapies, including antifibrotics and pulmonary hypertension-targeted treatments like OFEV and TYVASO/TYVASO DPI, rather than by increases in disease prevalence alone.
- The ILD pipeline is both extensive and diverse, featuring next-generation antifibrotic agents, immunomodulators, and lung-targeted delivery systems aimed at enhancing efficacy, safety, and phenotype-specific outcomes.
- Key mid- to late-stage candidates include taladegib (ENV-101), admilparant (BMS-986278), and seralutinib, alongside immune-targeting therapies such as belimumab (BENLYSTA) and efzofitimod.
- Emerging inhalation and localized delivery platforms, such as Treprostinil Palmitil Inhalation Powder (TPIP), L606, and AP01 (inhaled pirfenidone), are designed to optimize lung exposure while minimizing systemic side effects.
- Novel modalities like N115, a nonsteroidal nasal spray, further broaden the mechanistic landscape.
Learn more about the top companies working on IPF @ Interstitial Lung Disease Treatment Market
Interstitial Lung Disease Competitive Landscape
Some of the ILD drugs under development include Belimumab (BENLYSTA) (GSK), Treprostinil Palmitil Inhalation Powder (TPIP) (Insmed), Seralutinib (Gossamer Bio/Chiesi Farmaceutici), L606 (Treprostinil Liposome Inhalation Suspension) (Liquidia Corporation), Admilparant (BMS-986278) (Bristol-Myers Squibb), N115 (Nonsteroidal Nasal Spray) (Emphycorp), Efzofitimod (aTyr Pharma), Deupirfenidone (LYT-100) (PureTech Health), Taladegib (ENV-101) (Endeavor Biomedicines), AP01 (Inhaled Pirfenidone) (Avalyn Pharma), and others.
GSK’s Belimumab (BENLYSTA) is a B-lymphocyte stimulator (BLyS)–targeting inhibitor that binds to soluble BLyS, an essential factor for B-cell survival. As a fully human monoclonal antibody, it suppresses BLyS-driven prolonged survival of B cells, including autoreactive ones, and limits their maturation into immunoglobulin-secreting plasma cells.
Currently, belimumab is being assessed in advanced-stage (Phase II/III and Phase III) clinical trials for systemic sclerosis-associated interstitial lung disease (SSc-ILD) and connective tissue disease-associated ILD (CTD-ILD), highlighting the growing focus on B-cell-directed immunomodulatory therapies to maintain lung function and slow fibrotic progression.
Gossamer Bio/Chiesi Farmaceutici’s Seralutinib is an inhaled inhibitor of PDGFRα/β, CSF1R, and c-KIT, formulated as a dry powder for delivery via inhalation. It is being developed as a potential treatment for pulmonary arterial hypertension (PAH) and pulmonary hypertension linked to interstitial lung disease (PH-ILD).
Liquidia Corporation’s L606, developed in collaboration with Pharmosa Biopharm, is an investigational liposomal inhalation formulation of treprostinil designed for sustained release. This approach aims to enhance pulmonary delivery of treprostinil in patients with PAH and PH-ILD.
Insmed’s Treprostinil palmitil inhalation powder (TPIP) is a dry powder prodrug formulation in which treprostinil is esterified to a 16-carbon chain. It represents a potentially differentiated prostanoid therapy under investigation for PAH, PH-ILD, and other severe pulmonary conditions. Administered באמצעות a capsule-based inhalation device, TPIP remains an investigational product and has not yet received approval for any indication worldwide.
The anticipated launch of these emerging interstitial lung disease therapies are poised to transform the interstitial lung disease market landscape in the coming years. As these cutting-edge interstitial lung disease therapies continue to mature and gain regulatory approval, they are expected to reshape the interstitial lung disease market landscape, offering new standards of care and unlocking opportunities for medical innovation and economic growth.
To know more about late-stage ILD therapies, visit @ Interstitial Lung Disease Medication
Recent Developments in the Interstitial Lung Disease Market
- In March 2026, United Therapeutics reported that the Phase III TETON-1 trial assessing inhaled treprostinil achieved its primary endpoint in patients with idiopathic pulmonary fibrosis (IPF), indicating a notable improvement in lung function (FVC) and supporting the drug’s potential to extend beyond PH-ILD into more general ILD indications.
- In December 2025, PureTech Health announced the successful completion of the End-of-Phase II (EOP2) meeting with the US FDA regarding the development of deupirfenidone (LYT-100) for the treatment of IPF.
- In December 2025, US FDA approved nerandomilast (JASCAYD) tablets for the treatment of PPF in adults.
- In November 2025, Endeavor BioMedicines reported that the EMA granted PRIME designation to its investigational therapy taladegib for the treatment of IPF.
What is Interstitial Lung Disease?
Interstitial lung disease refers to a large and diverse group of lung disorders that primarily affect the interstitium, the delicate, lace-like network of tissue surrounding the air sacs in the lungs. In ILD, this tissue becomes inflamed and/or scarred, leading to reduced lung elasticity and impaired oxygen transfer into the bloodstream. Common causes include long-term exposure to environmental or occupational irritants, autoimmune diseases, certain medications, and infections, although in some cases the cause remains unknown, as seen in Idiopathic Pulmonary Fibrosis. Patients typically present with symptoms such as persistent dry cough, shortness of breath, and fatigue, which tend to worsen over time. While many forms of ILD are chronic and progressive, early diagnosis and appropriate management can help slow disease progression and improve quality of life.
Interstitial Lung Disease Epidemiology Segmentation
The interstitial lung disease epidemiology section provides insights into the historical and current interstitial lung disease patient pool and forecasted trends for the leading markets. According to DelveInsight’s estimates, in 2025, the US reported nearly 520,000 diagnosed prevalent cases of ILD.
The interstitial lung disease market report proffers epidemiological analysis for the study period 2022–2036 in the leading markets segmented into:
- Diagnosed Prevalent Cases of ILD
- Gender-specific Cases of ILD
- Type-specific Cases of ILD
- Treated Cases of ILD
Download the report to understand best-selling drugs for pulmonary fibrosis @ Interstitial Lung Disease Treatment Options
| Interstitial Lung Disease Market Report Metrics | Details |
| Study Period | 2022–2036 |
| Interstitial Lung Disease Market Report Coverage | 7MM [The United States, the EU-4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan] |
| Interstitial Lung Disease Market CAGR | 8.7% |
| Interstitial Lung Disease Market Size in 2025 | USD 6 Billion |
| Key Interstitial Lung Disease Companies | GSK, Insmed, Gossamer Bio, Chiesi Farmaceutici, Liquidia Corporation, Bristol-Myers Squibb, Emphycorp, aTyr Pharma, PureTech Health, Endeavor Biomedicines, Avalyn Pharma, United Therapeutics, Boehringer Ingelheim, Roche, and others |
| Key Interstitial Lung Disease Therapies | Belimumab (BENLYSTA), Treprostinil Palmitil Inhalation Powder (TPIP), Seralutinib, L606 (Treprostinil Liposome Inhalation Suspension), Admilparant (BMS-986278), N115 (Nonsteroidal Nasal Spray), Efzofitimod, Deupirfenidone (LYT-100), Taladegib (ENV-101), AP01 (Inhaled Pirfenidone), TYVASO/TYVASO DPI/TREPROST, YUTREPIA, JASCAYD, OFEV, ACTEMRA, and others |
Scope of the Interstitial Lung Disease Market Report
- Interstitial Lung Disease Therapeutic Assessment: Interstitial Lung Disease current marketed and emerging therapies
- Interstitial Lung Disease Market Dynamics: Conjoint Analysis of Emerging Interstitial Lung Disease Drugs
- Competitive Intelligence Analysis: SWOT analysis and Market entry strategies
- Interstitial Lung Disease Market Unmet Needs, KOL’s views, Analyst’s views, Interstitial Lung Disease Market Access and Reimbursement
Discover more about unmet needs in interstitial lung disease @ Interstitial Lung Disease Clinical Trials
Table of Contents
| 1 | Interstitial Lung Disease Market Key Insights |
| 2 | Interstitial Lung Disease Market Report Introduction |
| 3 | Interstitial Lung Disease (ILD) Market Overview at a Glance |
| 3.1 | Market Share (%) Distribution of ILD by Therapies in 2025 in the 7MM |
| 3.2 | Market Share (%) Distribution of ILD by Therapies in 2036 in the 7MM |
| 4 | Epidemiology and Market Methodology |
| 5 | Executive Summary |
| 6 | Key Events |
| 7 | Disease Background and Overview |
| 7.1 | Introduction |
| 7.2 | Signs and Symptoms |
| 7.3 | Causes |
| 7.4 | Risk factors |
| 7.5 | Pathophysiology of ILD Across Key Indications |
| 7.6 | Diagnosis |
| 7.7 | Treatment and Management |
| 7.8 | Diagnosis and Treatment Guidelines |
| 8 | Epidemiology and Patient Population |
| 8.1 | Key Findings |
| 8.2 | Diagnosed Prevalent Cases of ILD in the 7MM |
| 8.3 | The United States |
| 8.3.1 | Diagnosed Prevalent Cases of ILD in the United States |
| 8.3.2 | Gender-specific Cases of ILD in the United States |
| 8.3.3 | Type-specific Cases of ILD in the United States |
| 8.3.4 | Treated Cases of ILD in the United States |
| 8.4 | EU4 and the UK |
| 8.5 | Japan |
| 9 | Interstitial Lung Disease Patient Journey |
| 10 | Marketed Interstitial Lung Disease Therapies |
| 10.1 | Competitive Landscape: Marketed Drugs |
| 10.2 | Treprostinil (TYVASO/TYVASO DPI/TREPROST): United Therapeutics |
| 10.2.1 | Product Description |
| 10.2.2 | Regulatory Milestones |
| 10.2.3 | Other Development Activities |
| 10.2.4 | Summary of Pivotal Trial |
| 10.2.5 | Ongoing Clinical Activity |
| 10.2.6 | Analyst Views |
| 10.3 | Treprostinil (YUTREPIA): Liquidia Corporation |
| 10.4 | Nerandomilast (JASCAYD): Boehringer Ingelheim |
| 10.5 | Nintedanib capsules (OFEV): Boehringer Ingelheim |
| 10.6 | Tocilizumab (ACTEMRA): Roche |
| List to be continued in the final report… | |
| 11 | Emerging Interstitial Lung Disease Therapies |
| 11.1 | Competitive Landscape: Emerging Drugs |
| 11.2 | Belimumab (BENLYSTA): GSK |
| 11.2.1 | Product Description |
| 11.2.2 | Other Developmental Activities |
| 11.2.3 | Clinical Developmental Activities |
| 11.2.4 | Analyst View |
| 11.3 | Treprostinil Palmitil Inhalation Powder (TPIP): Insmed |
| 11.4 | Seralutinib: Gossamer Bio/ Chiesi Farmaceutici |
| 11.5 | L606 (Treprostinil Liposome Inhalation Suspension): Liquidia Corporation |
| 11.6 | Admilparant (BMS-986278): Bristol-Myers Squibb |
| 11.7 | N115 (Nonsteroidal Nasal Spray): Emphycorp |
| 11.8 | Efzofitimod: aTyr Pharma |
| 11.9 | Deupirfenidone (LYT-100): PureTech Health |
| 11.10 | Taladegib (ENV-101): Endeavor Biomedicines |
| 11.11 | AP01 (Inhaled Pirfenidone): Avalyn Pharma |
| List to be continued in the final report… | |
| 12 | Interstitial Lung Disease (ILD) Market – Seven Major Market Analysis |
| 12.1 | Key Findings |
| 12.2 | Interstitial Lung Disease Market Outlook |
| 12.3 | Conjoint Analysis |
| 12.4 | Key Market Forecast Assumptions |
| 12.5 | Market Size of ILD in the 7MM |
| 12.6 | Market Size of ILD by Therapies in the 7MM |
| 12.7 | The United States Interstitial Lung Disease Market |
| 12.7.1 | Total Market Size of ILD in the United States |
| 12.7.2 | Market Size of ILD by Therapies in the United States |
| 12.8 | EU4 and the UK Interstitial Lung Disease Market |
| 12.9 | Japan Interstitial Lung Disease Market |
| 13 | Key Opinion Leaders’ Views on ILD |
| 14 | Interstitial Lung Disease Market SWOT Analysis |
| 15 | Interstitial Lung Disease Market Unmet Needs |
| 16 | Interstitial Lung Disease Market Access and Reimbursement |
| 16.1 | United States |
| 16.2 | EU4 and the UK |
| 16.3 | Japan |
| 16.4 | Reimbursement Scenario in ILD |
| 17 | Bibliography |
| 18 | Interstitial Lung Disease Market Report Methodology |
Related Reports
Interstitial Lung Disease Epidemiology
Interstitial Lung Disease Clinical Trial Analysis
Interstitial Lung Disease Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, including clinical and non-clinical stage products, and the key interstitial lung disease companies, including AdAlta, Bristol-Myers Squibb, aTyr Pharma, Avalyn Pharmaceuticals, Beijing Continent Pharmaceutical, Regend Therapeutics, Reata Pharmaceuticals, FibroGen, PureTech Health, Bellerophon Pulse Technologies, OncoArendi Therapeutics, LTT Bio-Pharma, EmphyCorp, Genentech, Cudetaxestat, Boehringer Ingelheim, Prometheus Biosciences, HEC Pharm, Bayer, Insmed, Bristol-Myers Squibb, Avalyn Pharma, PureTech Health, Roche, Ark Biosciences, Novartis, Lung Therapeutics, Horizon, MediciNova, Endeavor BioMedicines, Pliant Therapeutics, Kadmon Pharmaceuticals, GenKyoTex, Taiho Pharmaceutical, Syndax Pharmaceuticals, Metagone Biotech, Galecto Biotech, CSL Behring and AstraZeneca, and others.
Pulmonary Hypertension associated with Interstitial Lung Disease Market
Pulmonary Hypertension associated with Interstitial Lung Disease Market Insights, Epidemiology, and Market Forecast – 2034 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, market share of the individual therapies, and key PH-ILD companies including Pharmosa BioPharm, Liquidia Corporation, Insmed, Roivant Sciences, Pulmovant, Halo Biosciences, Respira Therapeutics, Gossamer Bio, and others.
Connective Tissue Disease associated with Interstitial Lung Disease Market
Connective Tissue Disease associated with Interstitial Lung Disease Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key CTD-ILD companies, including Boehringer Ingelheim, Roche, Genentech, Bristol Myers Squibb, Novartis, Pfizer, Gilead Sciences, GlaxoSmithKline, AstraZeneca, Johnson & Johnson, Merck & Co., Sanofi, Eli Lilly and Company, AbbVie, Celgene (now part of Bristol Myers Squibb), Vertex Pharmaceuticals, Biogen, Amgen, Takeda Pharmaceutical Company, Daiichi Sankyo, Astellas Pharma, and others.
Progressive Fibrosing Interstitial Lung Disease Market
Progressive Fibrosing Interstitial Lung Disease Market Insights, Epidemiology, and Market Forecast – 2034 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, market share of the individual therapies, and key progressive fibrosing interstitial lung disease companies including Boehringer Ingelheim, Bristol-Myers Squibb, FibroGen, Hoffmann-La Roche, Pliant Therapeutics, United Therapeutics, Vicore Pharma AB, and others.
Systemic Sclerosis associated Lung Disease Market
Systemic Sclerosis associated Lung Disease Market Insights, Epidemiology, and Market Forecast – 2034 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, market share of the individual therapies, and key systemic sclerosis associated lung disease companies including Genentech Inc., Boehringer Ingelheim, Prometheus Biosciences Inc., Roche, Acceleron Pharma, Talaris Therapeutics, Kadmon Corporation, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.
CONTACT: Contact Us Shruti Thakur [email protected] +14699457679
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. Mango Bunch takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. Mango Bunch takes no editorial responsibility for the same.

